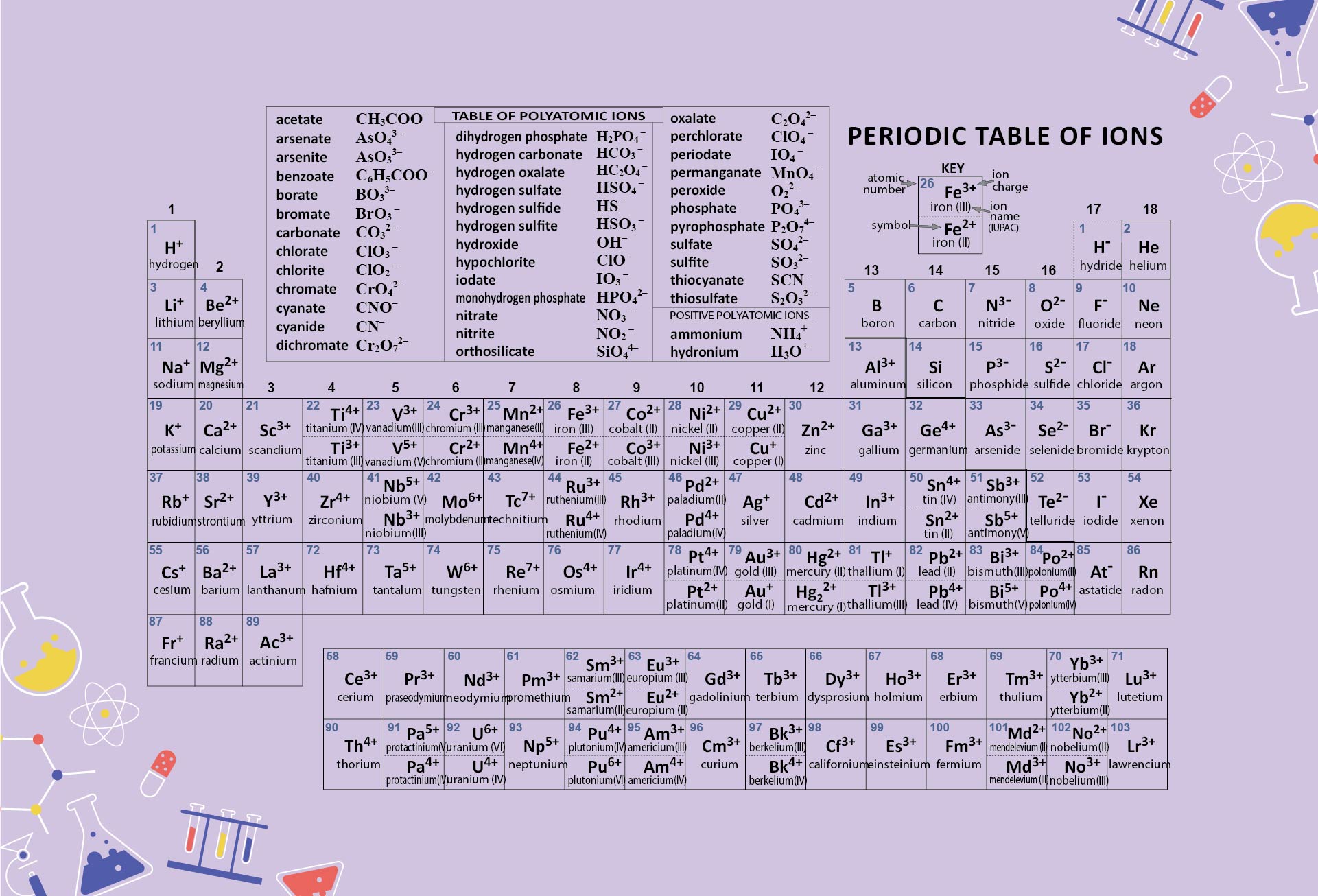
He used Alessandro Volta’s recently invented battery, which created electricity from a chemical reaction, to do the reverse. Though Dalton invented atomic theory, it was Berzelius who embedded it at the heart of the subject.Īnd Berzelius did more. And it was he who used these formulae to describe reactions: H 2SO 4 + Zn→ZnSO 4 + H 2 (sulphuric acid plus zinc becomes zinc sulphate plus hydrogen). It was he who combined those abbreviations with numbers, indicating the proportions involved, to make formulae for chemical compounds: H 2O (water), H 2SO 4 (sulphuric acid), NaCl (table salt). It was he who came up with the idea of the abbreviations that now occupy the periodic table’s rectangles. Jacob Berzelius, a Swede, furnished chemistry with its language. And it was quickly picked up by someone who, though less famous than Lavoisier, perhaps because of his grisly end, was arguably the greater man. Thus was the atom born.ĭalton based his system of relative atomic weights on hydrogen, the atoms of which he found to be the lightest. The Greek word for indivisible is “atomos”. 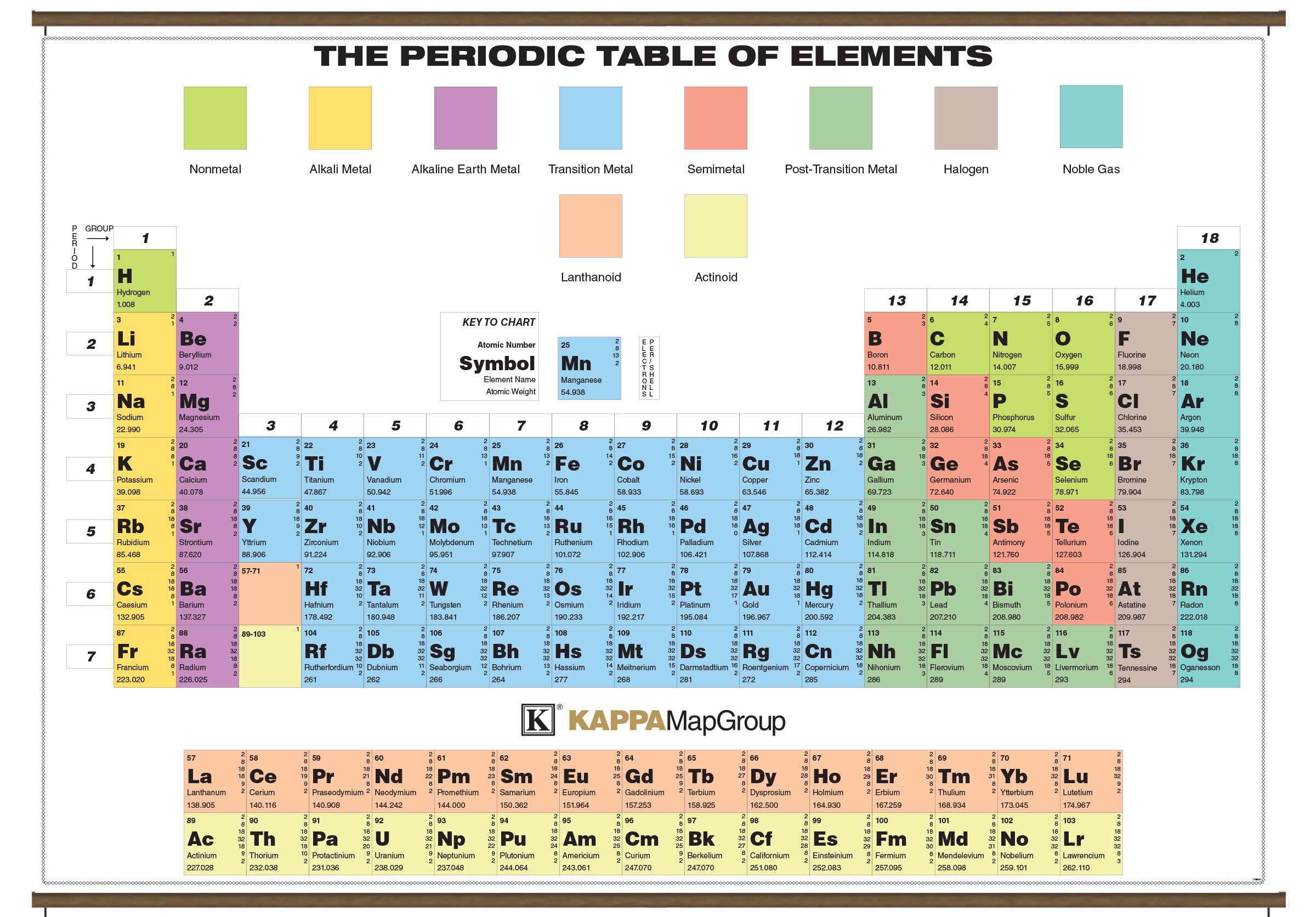
The simplest way to explain this-and indeed the way that Dalton lit upon-was to suppose each element to be composed of tiny, indivisible particles, all of the same weight. But during the first decade of the 19th century he took Proust’s concept and showed not only that elements reacted in fixed proportions by weight, but also that those proportions were ratios of small whole numbers. He made a modest living tutoring, but spent most of his energy on scientific research, including into colour-blindness, a condition still sometimes referred to as Daltonism. And he was English.ĭalton lived in Manchester, at a time when it was the world’s largest industrial city. The man himself was an ascetic, colour-blind Quaker. Dalton’s parents were so poor that he had been put to work at the age of ten. That insight had to wait for John Dalton, a man who was the polar opposite of the aristocratic bon vivant Lavoisier. From there, it might have been a short step for Proust to arrive at the idea of compounds being made of particles of different weights, each weight representing a specific element. It does not depend on that compound’s method of preparation. This law, published in 1794, the year of Antoine Lavoisier’s execution, states that the ratio by weight of the elements in a chemical compound is always the same. That fact established, another Frenchman, Louis-Joseph Proust, extended the idea with the law of definite proportions. Chemistry transforms the nature of substances, but not their total mass. The Lavoisiers’ careful measurements had discovered something now thought commonplace-the law of conservation of matter. It is little exaggeration to say that almost everything in modern science is connected, usually at only one or two removes, to the periodic table. Its rows and columns of rectangles, each containing a one- or two-letter abbreviation of the name of an element, together with its sequential atomic number, represent an order and underlying structure to the universe that would have astonished Lavoisier. It is now, though, a familiar feature of every high-school science laboratory. But the album was missing.Ĭreating that album, filling it and understanding why it is the way it is took a century and a half. /GettyImages-758302997-5c215e3846e0fb0001b8bf7b.jpg)
It was, avant la lettre, a stamp collection. What the list did not have was a structure. Others, like manganese, molybdenum and tungsten, were recent discoveries. Some, like gold, iron and sulphur, had been known since ancient days. Of those, 23-a fifth of the total now recognised-have stood the test of time. Lavoisier’s list of elements, published in 1789, five years before his execution, had 33 entries. But Lavoisier’s laboratory is as good a place as any to begin, for it was Lavoisier who published the first putatively comprehensive list of chemical elements-substances incapable of being broken down by chemical reactions into other substances-and it was Lavoisier and his wife Marie-Anne who pioneered the technique of measuring quantitatively what went into and came out of a chemical reaction, as a way of getting to the heart of what such a reaction really is. Where the story of the periodic table of the elements really starts is debatable.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
